Membrane-Mimicking Biosensors
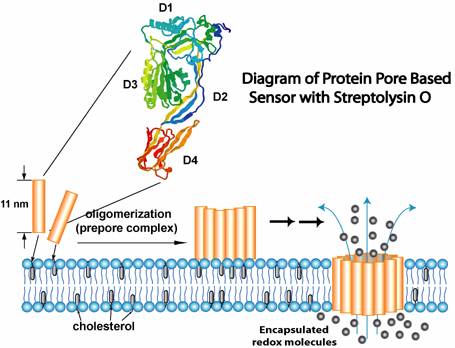
Biosensors are molecule-based devices that transduce a biochemical process or binding event into a measurable signal. We are interested in making sensors or sensor arrays that mimic nature's own biosensing mechanisms, which are widely observed in cells. Current effort has focused on detection of bacterial protein toxins that can bind to cell surface receptors and form channels or pores in the membrane. Many toxins are major determinants of bacterial virulence, and thus responsible for toxic activity and infection even without infestation by the microbes. Therefore, toxin sensors can offer effective tools for use in clinical diagnostics, food safety monitoring, epidemic control, and even counter-terrorism campaign. We study a variety of bacterial protein toxins and their interactions with receptors on bilayer membrane of vesicles. A recent project used lipid vesicles to encapsulate redox species to form a surface bound sensing interface through self-organizing process. The target protein toxin, streptolysin O (SLO) from Streptococcus pyogenes, interacts with membrane and form transmembrane pores of nanometer size, leading to release of redox molecules that enable amperometric signaling. We also investigate the use of PDMS microfluidic devices to build miniaturized chip sensors for multichannel analysis. The surface hydrophilicity of PDMS, which is critical to protein-based applications, has been optimized under aggressive study.
Additional interests include the quartz crystal microbalance (QCM) and SPR sensing techniques for spatially addressable micropatterned toxin sensors fabricated with supported bilayer membranes (SBMs). SBMs are formed by attaching a single lipid bilayer to a solid substrate either by physical interactions or by chemical bonds. They offer excellent film stability while a great degree of membrane fluidity can still be retained. Typical methods of fabrication involve fusion of bilayer vesicles, microcontact printing, or the Langmuir-Blodgett technique. We are exploring the building of SBMs with both the natural and synthetic lipids capable of inner crosslinking that further strengthens the membrane's robustness.
Functional Materials for Lipid Membrane Method Development
Traditionally, hydrophilic surfaces such as glass or unoxidized PDMS have been used for supporting lipid bilayers in sensor and biophysical experiment design. However, gold, which is the most common material in electrochemical and plasmonic techniques, inherently lacks the ability to support a stable and fluid lipid bilayer. We have developed multiple techniques for creating nanoscale layers of glass on gold surfaces that allow for in situ supported lipid bilayer assays to be performed. Our layer-by-layer deposition/calcination protocol is largely based on wet chemistry and may be performed without expensive cleanroom or vacuum equipment (down to ca. 2 nm SiO2). Alternatively, plasma-enhanced chemical vapor deposition may be applied for bulk material processing (down to 4 nm SiO2). With these, we have created multiplexed lipid bilayer arrays for SPR imaging, electrochemistry, and fluorescence. Further work is focusing on improving arraying protocols and expanding these methods to complex lipid environments.
Applications of Synthetic Cavitand Receptors (Collaboration with Prof. Richard Hooley)
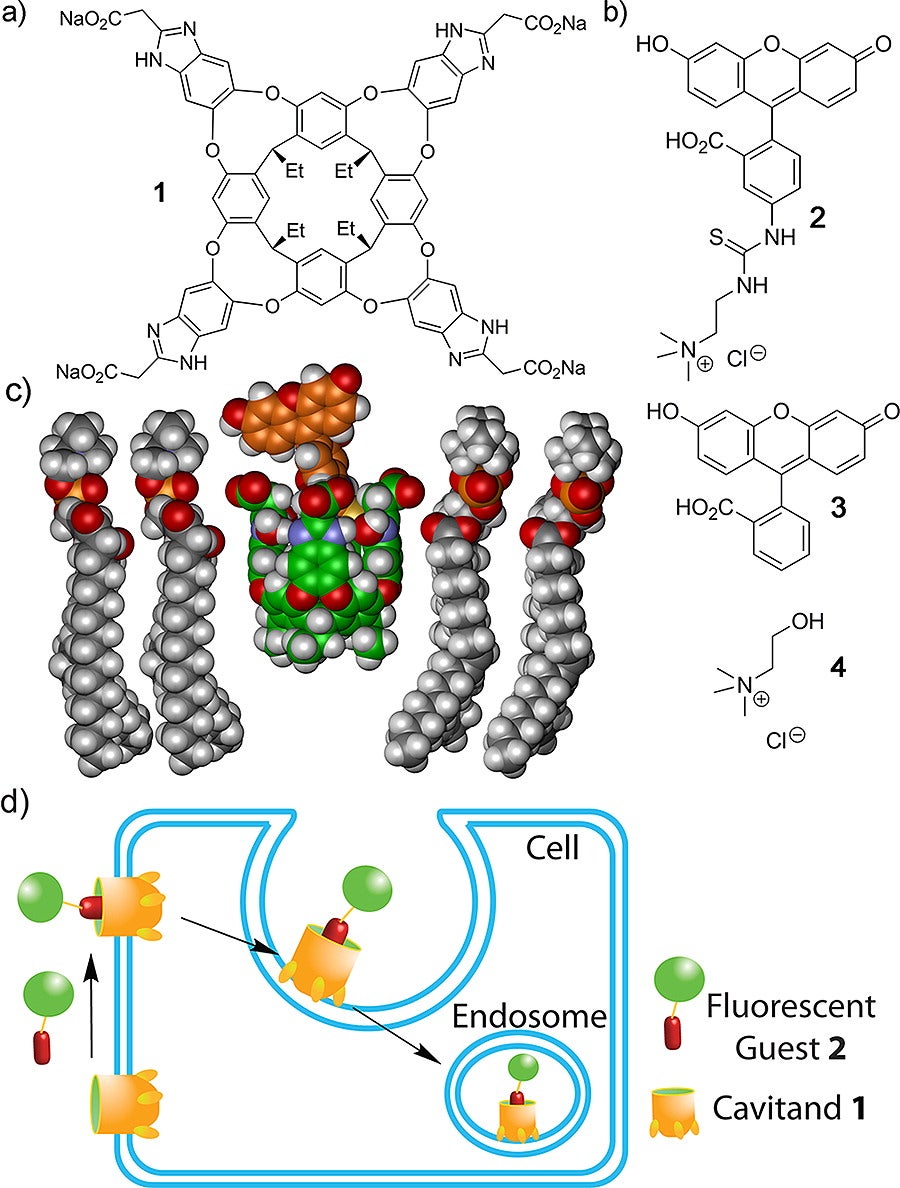
From the Hooley Group Website: Synthetic host molecules non-covalently bind substrates with the correct size and shape complementarity. We study deep cavitands that can be incorporated in supported bilayer membranes, while retaining their host properties, allowing reversible binding and real-time sensing of a variety of biologically important analytes. Real-time analysis of the process is performed via surface plasmon resonance spectroscopy. This system provides a system, flexible architecture for reactions and molecular recognition at a bilayer interface, controlled solely by cavitand:guest interactions in a truly biomimetic environment. This can be explored for the in situ creation of polymer films atop the lipid bilayer, a process only possible when mediated by the membrane-bound cavitand. Our system is unique: the process occurs at the bilayer itself, under mild conditions and the process can be reversed, as the polymer is connected to the bilayer through non-covalent molecular recognition with the cavitand hosts. The ultimate goal is to use the recognition system for a study of cell adhesion and sorting via the membrane-bound synthetic receptors or functionalized polymer domains on a supported lipid membrane interface.